VSRF LIVE 100th Episode Tomorrow: Time To Sue Pfizer?
With Covid Litigation Attorneys Tricia Lindsay and Jeff Childers
VSRF LIVE
Thursday, October 26
7pm Eastern | 4pm Pacific
Live stream on Rumble
Important Note: VSRF Live is exclusively on Rumble - NO MORE ZOOM

Join us for our 100th episode of VSRF LIVE this Thursday. We will directly address the controversy surrounding my tweet about the SV40 Plasmidgate adulteration and Pfizer litigation, which sparked a fierce debate. I will be speaking with two exceptional legal minds in the COVID litigation arena, Tricia Lindsay and Jeff Childers, both distinguished speakers at the first Covid Litigation Conference and our upcoming Covid Litigation Conference II.
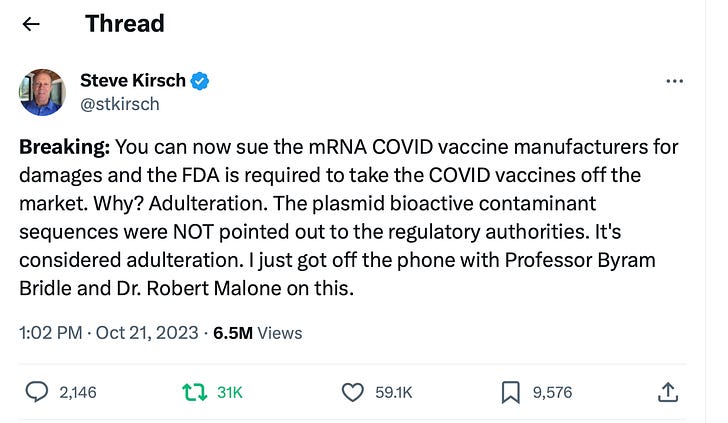
Is the SV40 adulteration, which Pfizer did not disclose to regulators in America, a routine manufacturing technique (as Alex Berenson argues), or does it provide the grounds to vitiate the PREP ACT and finally hold Pfizer accountable for COVID-19 vaccine injuries and deaths?

Moreover, we will not just be addressing this question from a technical perspective. We will also address it by discussing the pressing concerns faced by those affected by vaccine injuries and losses. What steps can they take in light of these revelations? How can we offer support to the lawyers passionately working in this space? These are the critical questions we will answer tomorrow night on VSRF LIVE as we build pathways toward justice and accountability.

At VSRF, we are dedicated to this cause, and one of our initiatives is the upcoming Covid Litigation Conference II, scheduled for March 7-8th in Las Vegas. This conference is not just an event; it's a platform where legal minds unite to strategize, collaborate, and initiate legal actions against unjust Covid policies. I urge you to attend and encourage any attorneys you know to join us. The more Covid-related lawsuits we initiate, the harder it becomes for these policies to resurface in the future
However, to fuel initiatives like the Covid Litigation Conference II, VSRF needs your support. We will air our 100th VSRF Live episode this week, a testament to the journey we have embarked on together. Your backing has brought us this far, and your continued support is vital for our continued success. Please consider contributing a monthly donation to keep VSRF running. Your support is not just a contribution; it's an investment in the fight against the corrupt elite, ensuring that we can keep the lights on and continue our mission to reclaim our rights and freedoms.
You can donate at VacSafety.org/donate
Thank you for being an integral part of this movement. Together, we will win.
Join us tonight and bring a friend. Please also share widely! Just re-post any of these links:


I like all the interviews but I especially like the ones about taking action! We're all saying "What can we do?" Here's one answer. Plus Tricia Lindsay is a joy to listen to.
I am SO tired! It has been three years of this crap.